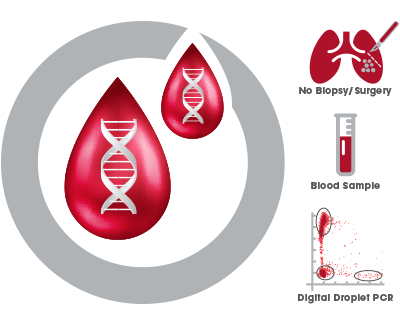
ctDNA testing provides a rapid, minimally invasive blood testing alternative to traditional biopsy or resection tissue analysis of EGFR T790M mutation status in non-small cell lung adenocarcinoma.
This test is ideally suited for testing blood plasma or cerebrospinal fluid for:
- EGFR T790M drug-resistant mutation detection at initial diagnosis or recurrence of non-small cell lung adenocarcinoma.
- Quantitative, highly-sensitive (0.5–
< 0.01% mutant allele frequency*) monitoring of therapeutic response or disease progression in patients eligible for EGFR T790M-specific TKIs.
* based upon the amount of amplification DNA, the limit of detection ranges from 0.5% to less than 0.01% mutant alleles.
Typical Testing Strategy
In patients with EGFR gene mutations who are treated with early-generation TKIs, consider monitoring for development of an acquired EGFR T790M mutation.
- Blood plasma
- Serial quantitative testing
- Guides clinical treatment decisions about acquired resistance to early-generation TKIs and decision to switch to EGFR T790M-specific TKIs.
- Monitors response to therapy in patients taking EGFR T790M-specific TKIs.
- Serial quantitative testing
- Cerebrospinal fluid
- Serial quantitative testing for patients with isolated NSCLC brain metastases and undetectable blood plasma levels of EGFR T790M mutation.
- Guides clinical treatment decisions about switching to EGFR T790M-specific TKIs.
- Monitors response to therapy.
Additional Resources
- Spotlight on Testing: Circulating cell free tumor DNA and EGFR T790m testing Mary Bronner, MD, Division Chief, Anatomic Pathology and OncologyCarl R. Kjeldsberg, Presidential Endowed Professor of Pathology, University of Utah School of Medicine
- Breakthrough Technology Spurs New Test: for Non-Small Cell Lung Cancer Patients Wade Samowitz, MD, Medical Director, Solid Tumor Molecular Diagnostics and Histology, Staff Pathologist, Anatomic Pathology
- Revolutionizing Treatment for Cancer Care: New Circulating Cell-Free Tumor DNA (ctDNA) Technology Mary Bronner, MD, Division Chief, Anatomic Pathology and Oncology